And what you've all been waiting for
Here's another kind of possible problem: Using the equation governing the flow of sugar across a membrane from the following equation
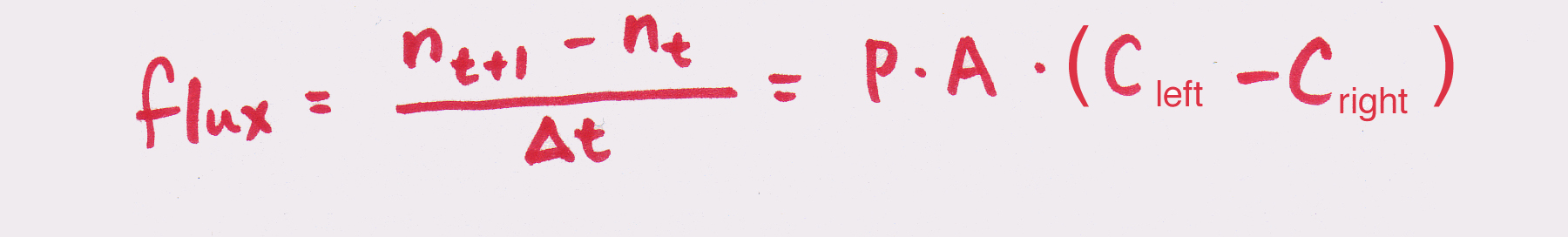
And given that
P= 0.02 cm/sec
the area of the membrane between the two solutions is 2 cm2
Solution left side of the membrane = 0.4M
Solution right side of the membrane = 0.1M
What is the rate of diffusion across the membrane for the following concentrations? This kind of problem requires straightforward plug-and-chugging...
0.02cm/sec x 2 cm2 x (0.4M sugar - 0.1M sugar)
= 0.012 moles/second
What would happen if we doubled the permeability constant?
What if we doubled the area of the membrane?
What if we doubled the concentration on the left?
Copyright University of Maryland, 2007
You may link to this site for educational purposes.
Please do not copy without permission
requests/questions/feedback email: mathbench@umd.edu