Well, not quite that easy
Avogadro had in mind the "standard" form of carbon which has 6 protons and 6 neutrons. But some carbon atoms have an extra neutron — in fact this is what allows us to do carbon dating ... but that's another different story.
So, when you take the extra-big carbons into account, the average weight of many carbon atoms, as found in nature, is slightly more than the weight of the 6-neutron kind.
And therefore, a mol of carbon found in nature actually weighs 12.011 g. Technically, the number 12.011 has no units and is called the "atomic mass" of the element. If you give this number the units of grams/mol, you now have the "molar weight" of the atom.
(Important note
Do not confuse molar weight with molarity!! Molarity (also called "molar concentration") has to do with how many moles of the substance you put into a solution. Molar weight has to do with how much a given amount of a particular kind of molecule weighs.
End of important note)
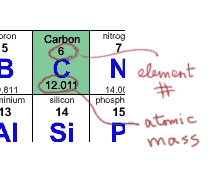
In fact all of this information is nicely summarized on a periodic table. For example, here is the entry for carbon:
| If you turn on javascript, this becomes a rollover |
This neat little trick completely avoids the awful calculation we were faced with before:
.0000000000000000000000664 g * 602,200,000,000,000,000,000,000 = who cares, because now we can just look up helium on the periodic table
When you need to figure out the molar weight of a compound, always make sure you have one of these tables with the atomic mass of each element. The element number will probably be at the top (6, in the case of carbon) and the atomic mass down below. In any case, you know that the element number (the number of protons) should increase by one per cell as you move across the table. The atomic mass is always at least double the element number, because there are always at least as many neutrons as protons. Large elements like Uranium have extra neutrons, so their atomic masses are more than double their element numbers.
Gotcha ...
— yes, I know, a few people out there are dying to tell me that the atomic mass of Hydrogen is NOT double its molecular number, because it has no neutrons. Well done.
Copyright University of Maryland, 2007
You may link to this site for educational purposes.
Please do not copy without permission
requests/questions/feedback email: mathbench@umd.edu