Our equations are ready
So let's add a linear term for area of the membrane. Hmm, let's call it A, for area. No one can accuse mathematicians of getting too fancy with their names. So here are our final (for now) equations:
| Continuous version |
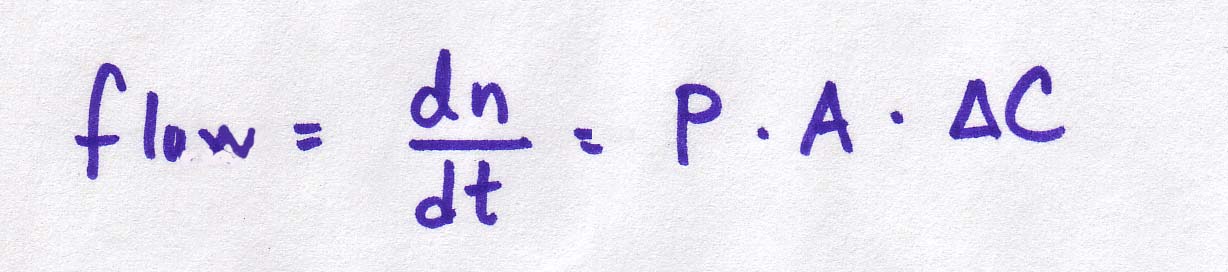
|

|
|---|---|---|
| Discrete version |
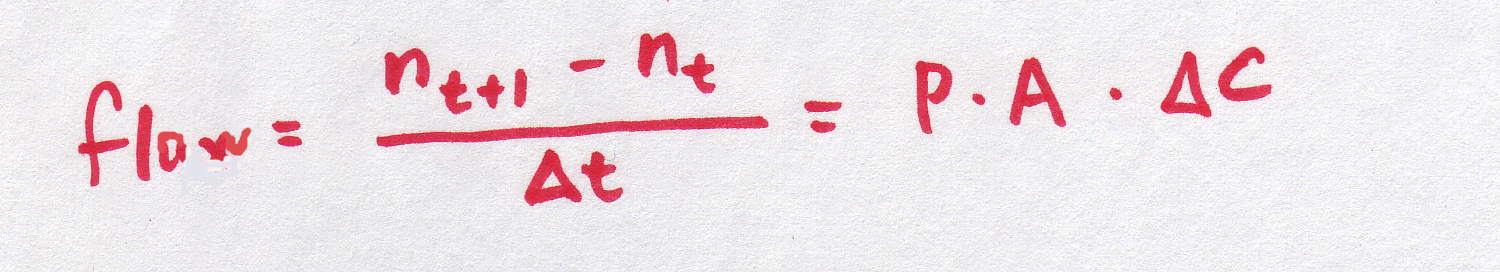 |
In order to do any calculations, we will have to get a handle on the actual values of these variables.
That means we need to be able measure concentration. You might think this
is pretty easy. For example, if you put 100 grams of sugar in 900 grams of
water, you will have a solution that is 10% sugar by weight. And you probably
don't want to try to drink it. (Honest, even Mountain Dew is only about 8%
by weight). 
However, unfortunately, diffusion doesn't care about weight, it cares (if a process can be said to care) about particles . How many particles of sugar were in that 100 grams?
 Before you get out a magnifying glass and start counting sugar grains, let's
be clear about what a particle is: as a first approximation, a particle is
a molecule. So, you need to know the molecular weight of sugar C6H12O6.
Before you get out a magnifying glass and start counting sugar grains, let's
be clear about what a particle is: as a first approximation, a particle is
a molecule. So, you need to know the molecular weight of sugar C6H12O6.
If you remember how to calculate molecular weight, go ahead and do it now. If not, the next page is just for you...
Copyright University of Maryland, 2007
You may link to this site for educational purposes.
Please do not copy without permission
requests/questions/feedback email: mathbench@umd.edu