Measuring Movement Using Flux
Since we are interested in a specific process (movement), we need a good way to measure that process. As scientists, we love to measure things. In fact, if we can't measure something, we're probably not very interested in it. As it turns out, there are all sorts of ways to measure movement, but we are going to focus on the quantity known as flux.
Flux is an unusual concept for many people because its ordinary (conversational) meaning is rather different from its scientific meaning. For most people (meaning the two people we just asked!) flux means changes in conditions, similar to “ebb and flow” or “fluctuations”. But its scientific meaning is “the net rate at which particles move through a certain area” .
That is, flux is the net movement of particles across a specified area in a specified period of time. The particles may be ions or molecules, or they may be larger, like insects, muskrats or cars. The units of time can be anything from milliseconds to millennia. Here are some examples:
 |
 |
 |
The number of cars that pass through a toll booth
every day. |
The net number of muskrats that cross a fence every
hour. |
The net number of salt ions that pass out of a cell
membrane every minute. |
Flux is NOT the same thing as velocity or speed, which are measured in the units of distance per time, rather than number per time. Individual oxygen molecules may be moving very fast, but since they are going in a variety of directions (they are undirected), there will be no net movement
Likewise, flux is also NOT the same thing as density or concentration, which are measured as particles per volume. A cell may be chock-full of oxygen, but if none of the oxygen molecules are going anywhere, there is no flux. Or there may be only a little oxygen, but what is there is
Finally, movement itself is not enough. If (like some cats I know) you continually go in and out of the same door, we wouldn’t say there was high flux through the door. Instead, flux is a measure of NET movement.
In one minute 0.0019 moles of water move into a cell and 0.0012 moles move out of the cell. One mole of water weights 18 g, and the volume of the cell is 13 µm3, and its surface area is 28 µm2. What is the flux of water molecules into the cell?
(To make this problem interactive, turn on javascript! ...also, some elements may work better in Firefox)
- I need a hint ... : All you need to know is the net rate of movement and the area. The rest of information is unnecessary.
- ...another hint ... : Net movement
= movement in - movement out = 0.0007 moles - ...another hint ... : Net rate of movement
= 0.0007 moles / 1 minute
= 0.0007 moles/min - ...another hint ... : Don't forget that flux is per area!
I think I have the answer: Net rate of movement / area = (0.0007 moles/min) / 28 µm2 = 0.000025 moles/min/ µm2
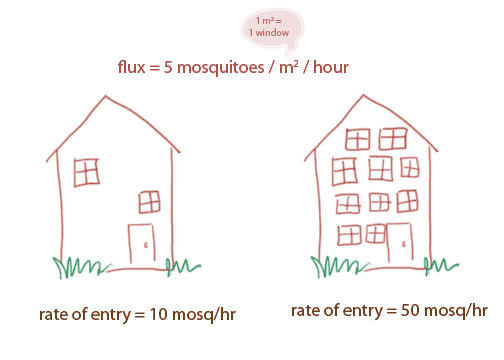
Notice that we could use the flux to calculate the actual amount of molecules going in an out of an area. Here's an analogy: let's say I know the net flux of mosquitoes moving into my house: 5 mosquitoes per hour per square meter window. This is flux, because it is particles (mosquitoes) per time (hours) per area (meter square windows).
If I want to know how many total mosquitoes enter my house per hour, I need to know one more thing: how many windows my house has!! A house with 2 windows will have far fewer mosquitoes entering than a house with 10 windows!
(mouse over the image for how the rates were calculated!)
We can generalize to say that
rate = flux * area
which is really just a rearrangement of
rate / area = flux.
You should be able to figure it out in either direction. On the problem above, I could have said
The flux of water into a cell is 0.000025 moles second per micron squared, and the surface area of the cell is 28 microns squared. What is the total rate at which water is moving into the cell?
Answer: 0.000025 moles / second / μm2 * 28 μm2 = 0.0007 moles/min
So the only difference between flux and rate is that flux is looking at an area of a specific size.
Copyright University of Maryland, 2007
You may link to this site for educational purposes.
Please do not copy without permission
requests/questions/feedback email: mathbench@umd.edu