Overall dilution
Now that you are very comfortable with dilution factors... what happens if you dilute and then dilute again? For example, we could take super-strong cofee, dilute it by 1/5, then dilute THAT by 1/10. So,
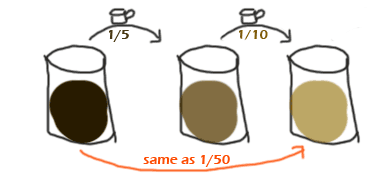
 Here's the rule:
Here's the rule:
To find the overall (or total) dilution factor,
simply multiply the dilution factors for each step.
Once again, here's an applet to practice finding the total dilution scheme, regardless of how the dilution scheme is expressed -- as directions, as fractions, or as decimals.
Remember, keep going until you're very comfortable with the calculations... Oh, and do yourself a favor, use a calculator!!
Given the following series of dilutions,
what is the total dilution factor?




| As a Fraction: 1 / | |
| As a Decimal: | |
|
feedback here
| |
Copyright University of Maryland, 2007
You may link to this site for educational purposes.
Please do not copy without permission
requests/questions/feedback email: mathbench@umd.edu